Definition:
In chemistry, “aq” stands for “aqueous.” It indicates that a substance is dissolved in water, forming a solution. The abbreviation comes from the Latin word “aqua,” meaning water. Chemists place (aq) after a chemical formula to show that the compound exists in a water-based solution rather than as a solid, liquid, or gas.
Water is often called the universal solvent, and in chemistry, it plays a starring role in countless reactions. But have you ever noticed the small letters “aq” next to a chemical formula and wondered what they mean? That tiny abbreviation carries big information: it tells you that the substance is dissolved in water, forming an aqueous solution ready to react.
For example:
- NaCl (aq) → Sodium chloride dissolved in water
- HCl (aq) → Hydrochloric acid in aqueous solution
- AgNO₃ (aq) → Silver nitrate dissolved in water
The (aq) symbol helps scientists quickly understand the state of a substance in a chemical reaction, which is important for predicting how reactions occur.
What Does AQ Mean in Chemistry?
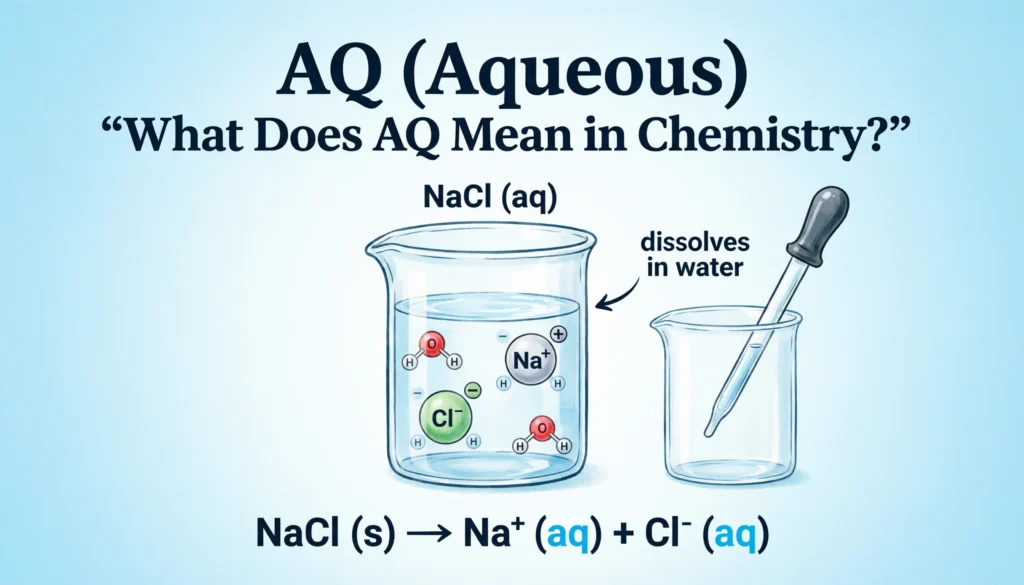
In chemical equations and laboratory notes, the abbreviation (aq) tells us that a compound is dissolved in water and exists as ions or molecules within a solution.
When a substance dissolves in water, its particles separate and spread throughout the liquid. This process allows many chemical reactions—especially ionic reactions—to occur efficiently.
For instance:
NaCl (s) → Na⁺ (aq) + Cl⁻ (aq)
This equation shows that solid sodium chloride dissolves in water and separates into sodium and chloride ions.
Scientists include (aq) to clearly describe:
- The physical state of a substance
- The reaction environment
- How substances interact in solution
Without this notation, interpreting a chemical equation would be much harder.
Origin of the Term AQ
The abbreviation aq originates from the Latin word “aqua,” meaning water.
Latin has historically influenced scientific terminology, especially in fields like chemistry, biology, and medicine. Because of this tradition, chemists adopted aq as the shorthand for aqueous solutions.
Why Latin Terms Are Used in Science
Several reasons explain why Latin-based terms remain common:
- Universality: Scientists worldwide understand standardized terms
- Historical tradition: Early scientific texts were written in Latin
- Clarity: Short abbreviations like (aq) simplify complex equations
Even today, when chemists write reactions, they rely on these traditional abbreviations for clarity and efficiency.
Why AQ Is Important in Chemical Reactions
Understanding (aq) is crucial because the state of matter affects how reactions occur.
Water acts as an excellent solvent, allowing ions and molecules to move freely. This mobility enables many reactions such as:
- Acid–base reactions
- Precipitation reactions
- Electrochemical reactions
- Neutralization reactions
Key Reasons Chemists Use (aq)
- To show a compound is dissolved in water
- To indicate free-moving ions in solution
- To help predict reaction outcomes
- To describe reaction conditions accurately
Without identifying the state of substances, chemical equations would lose essential information.
Common Chemical States Compared
In chemistry, substances are often labeled with abbreviations showing their physical state.
| Symbol | Meaning | Description | Example |
|---|---|---|---|
| (s) | Solid | Substance in solid form | NaCl (s) |
| (l) | Liquid | Pure liquid state | H₂O (l) |
| (g) | Gas | Gaseous state | CO₂ (g) |
| (aq) | Aqueous | Dissolved in water | NaCl (aq) |
This notation allows chemists to quickly visualize reaction conditions.
Examples of AQ in Chemical Equations
Below are common reactions where (aq) is used.
Example Table: AQ in Chemistry
| Chemical Equation | Explanation |
|---|---|
| HCl (aq) + NaOH (aq) → NaCl (aq) + H₂O (l) | Acid-base neutralization reaction |
| AgNO₃ (aq) + NaCl (aq) → AgCl (s) + NaNO₃ (aq) | Precipitation reaction forming solid silver chloride |
| NaCl (s) → Na⁺ (aq) + Cl⁻ (aq) | Dissolution of salt in water |
| CuSO₄ (aq) + Zn (s) → ZnSO₄ (aq) + Cu (s) | Single displacement reaction |
These examples show how (aq) signals substances that are dissolved in water and actively participating in reactions.
How AQ Appears in Laboratory Work
In real-world chemistry labs, scientists frequently work with aqueous solutions.
Common laboratory processes involving (aq) include:
- Preparing solutions
- Mixing reagents in water
- Performing titrations
- Observing precipitation reactions
- Measuring conductivity
Water is often used because it:
- Dissolves many compounds
- Is safe and inexpensive
- Allows ions to move freely
- Supports many chemical reactions
As a result, (aq) appears in countless lab experiments and textbooks.
Real-World Applications of Aqueous Solutions
Aqueous chemistry plays a major role in everyday life and industrial processes.
Everyday Examples
- Salt dissolved in water for cooking
- Sugar dissolved in tea or coffee
- Cleaning solutions containing dissolved chemicals
- Electrolyte drinks containing dissolved salts
Industrial Applications
- Pharmaceutical manufacturing
- Water treatment processes
- Battery chemistry
- Food processing
- Chemical manufacturing
Because water is such a versatile solvent, aqueous reactions are among the most common chemical processes in the world.
Comparison: AQ vs Other Chemical Abbreviations
Chemistry uses several abbreviations to describe states and conditions.
| Abbreviation | Meaning | Example | Use |
|---|---|---|---|
| (aq) | Aqueous | NaCl (aq) | Dissolved in water |
| (s) | Solid | CaCO₃ (s) | Solid substance |
| (l) | Liquid | H₂O (l) | Liquid form |
| (g) | Gas | O₂ (g) | Gas phase |
| Δ | Heat applied | CaCO₃ → CaO + CO₂ | Heating reaction |
Understanding these symbols helps students and scientists interpret chemical equations quickly and accurately.
Does AQ Have Other Meanings?
Although aq mainly means aqueous in chemistry, it can have other meanings in different fields.
| Field | Meaning |
|---|---|
| Chemistry | Aqueous (dissolved in water) |
| Business | Account |
| Internet slang | “As quoted” or shorthand for something similar |
However, in scientific writing, aq almost always refers to aqueous solutions.
Tips for Understanding Chemical State Symbols
If you’re learning chemistry, recognizing these symbols will make equations easier to read.
Helpful Tips
- Always check the state symbol after a chemical formula
- Remember (aq) = dissolved in water
- A reaction involving (aq) often includes ions
- Precipitation reactions produce a solid (s) from aqueous solutions
With practice, these symbols become second nature when reading chemical equations.
FAQs
What does (aq) mean in a chemical equation?
(aq) means the substance is dissolved in water, forming an aqueous solution where ions or molecules move freely.
What is an aqueous solution in chemistry?
An aqueous solution is a mixture where water acts as the solvent, dissolving another substance called the solute.
Why is aq used in chemical equations?
Chemists use (aq) to indicate the physical state of a compound, which helps explain how reactions occur.
What does NaCl (aq) mean?
NaCl (aq) means sodium chloride dissolved in water, forming sodium and chloride ions in solution.
What is the difference between (aq) and (l)?
- (aq) = dissolved in water
- (l) = pure liquid form
For example:
- HCl (aq) → hydrochloric acid dissolved in water
- H₂O (l) → liquid water
Can all substances become aqueous?
No. Only substances that dissolve in water can form aqueous solutions. Some compounds are insoluble.
What reactions commonly use aqueous solutions?
Common reactions include:
- Acid-base reactions
- Precipitation reactions
- Redox reactions
- Neutralization reactions
Why is water used as a solvent in chemistry?
Water is widely used because it:
- Dissolves many substances
- Is chemically stable
- Is safe and inexpensive
- Supports many types of reactions
Conclusion
The abbreviation (aq) is a small symbol with a very important meaning in chemistry. It tells us that a substance is dissolved in water and exists as an aqueous solution.
Understanding aq helps students and scientists interpret chemical equations, predict reactions, and understand how substances behave in water.
By learning these basic symbols, you can read chemical equations more confidently and understand how reactions happen in real-world science.
Discover More Related Articles:
- “I Am” Mean in the Bible: God’s Eternal Name Explained in 2026
- 10 Mean in the Bible: Spiritual Lessons in 2026

Amanda Lewis is a professional content writer and word-meaning researcher who specializes in explaining definitions, slang, abbreviations, and modern language terms. She writes for WordNexy.com, where she creates clear, accurate, and reader-friendly articles to help users understand word meanings and proper usage. Her work is especially useful for students, writers, and online readers seeking quick and reliable explanations.